Selecting Power Supplies for Production in the Face of Supply Chain Challenges
April 9, 2020 by Bruce Rose -
7 Minute Read
Last updated August 8, 2023

As everyone is aware, we were introduced to a different world during the peak period of COVID-19 due to many issues. As an example, we regularly heard news of companies and individuals working to provide long-term solutions to supply chain problems. Often mentioned in the news was the shortage of medical equipment during the pandemic, and the many companies that looked to quickly manufacture equipment to address those shortages.
For various reasons, disruptions like those present during the COVID-19 crisis will continue to happen periodically in the future. In addition to medical issues, future disruptions may be caused by political activities, earthquakes, fires, environmental changes, or other many other similar types of events. This blog will review the reasons why long lead times happen and discuss activities that may serve as a partial solution to the shortage issues.
Build to Order and Just in Time
Perhaps the biggest reason that electronic product suppliers cannot quickly ramp up production of tens or hundreds of thousands of units in times of need is due to the efficient business model that has steadily developed over the past decades. The electronics industry is only one of many market segments that have adopted the build to order (BTO) and just in time (JIT) business models to improve efficiency. These techniques work well when companies can receive long term forecasts and thus predict market needs beyond just the near future. Unfortunately, BTO and JIT techniques do not serve well when there is a severe or near-term disruption in business plans.
The BTO and JIT models extend through manufacturers of products to the suppliers of these manufacturers. In an efficient system all the suppliers in the chain are operating at or close to capacity and producing components that have forecasted requirements in place. Addressing near term and unexpected increased demands often requires reallocating existing work in progress (WIP) from the previously forecasted demand to the new un-forecasted demand. In the case of certified components such as those used for medical applications, the WIP would be re-allocated from one medical product to a different medical product and thus possibly not addressing the disruption causing the increased demand.
Among other functions, component distributors serve as a buffer for variances in BTO and JIT but the distributors have also developed practices to stock only the amount of inventory that is typically required by their customers. Purchase requests due to large surges in demand, as seen with the COVID-19 event, exceed the inventory holdings of the distributors and they must then place orders in the BTO and JIT queues of their suppliers.
Enabling Rapid High Volume Production of Existing Medical Products
During the COVID-19 event, there was a desire to not only create new designs but to also increase the production quantity of existing medical product designs quickly. In the existing designs the internal components had already been selected and thus the option was to either acquire more of the specified components or to find acceptable substitute parts (known as “crosses”). The challenge in selecting acceptable substitute parts is that they must be equivalent in form, fit, and function.
- Form: Being equivalent in form refers to having the same physical dimensions.
- Fit: Refers to the mechanical and electrical connections.
- Function: The component operates in a similar manner as does the original component.
We will discuss power supplies as an example of a component that may be in short supply and thus require a substitute. Some power supplies may serve as appropriate crosses to those from other manufacturers, but the requirements of the power supply function need to be carefully specified and evaluated by the customer. The original power supply data sheet should be reviewed to determine which specifications are required and which specifications are not important and then to ensure that the required specifications are met. Typically, the complete list of specifications between any two power supplies will not be identical. Due to the differences in specifications, it will be most efficient for the new vendor to confirm that their supply will work in the application with significant cooperation from a customer engineer who is familiar with the application.
Regulatory Approvals and Waivers
Medical products provide a good example of the challenges that can be encountered when creating new components or products. Medical grade electronic power supplies require regulatory testing and approval before they can be sold. Designing or modifying a power supply or opening a new manufacturing facility are all activities that may initiate the requirements for regulatory testing and approval activities; these activities often take multiple weeks to be completed. In addition to testing of the components, the final medical product will also be required to undergo regulatory testing and approval processes.
Design Philosophy for Enabling Rapid Production
Knowing that quickly acquiring high volumes of components can be an issue (due to unforeseen circumstances like a pandemic), there are design decisions one can make to minimize these challenges. A high-level design philosophy might be to design the product so it consists of independent modules rather than as a tightly integrated platform. The power supply for a product is an example of a sub-system that can be constructed from independent modules. While substitute modules will need to be functionally equivalent and may need to be form and fit equivalent, the components within the modules will not need to be form, fit, and functionally equivalent. The following are examples of power module architectures incorporating different internal components but all achieving the system requirements:
- Module A: a single ac-dc supply with all of the required output voltages produced by the one supply.
- Module B: multiple ac-dc supplies where each of the supplies generate some of the required output voltages and the combination produces all of the required voltages.
- Module C: a single ac-dc supply with multiple dc-dc converters to produce the required output voltages.
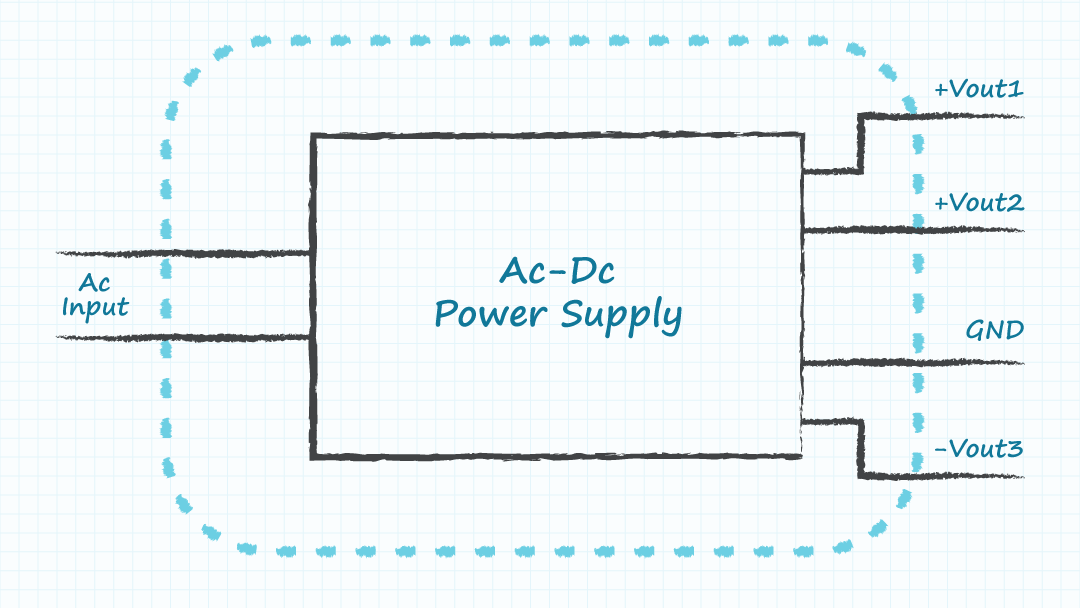
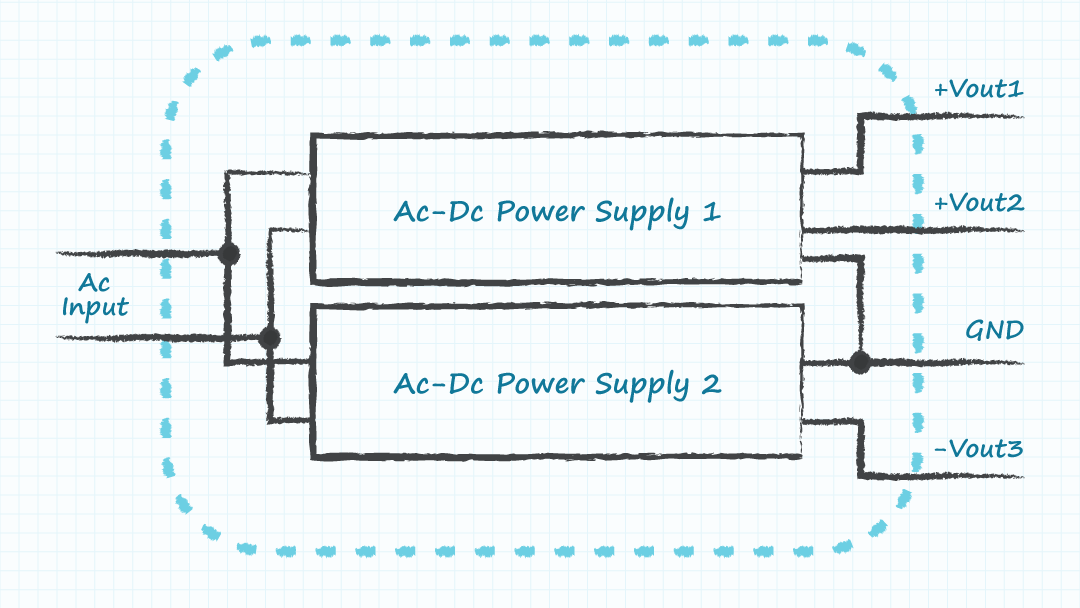
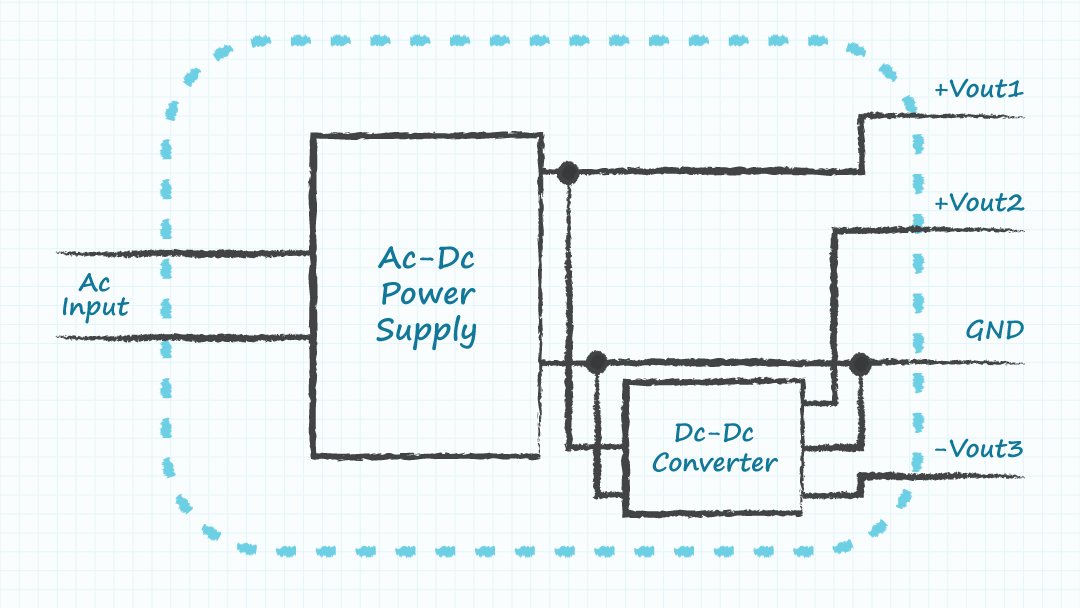
Including wall mount or desktop external power supplies within a module can also be an option for ac-dc supplies. The ability to acquire sufficient components to build lower volumes of many equivalent modules could enable high volume production of the final product by using many versions of the low volume modules. It is recognized that the cost, size, and performance of the system may be compromised by a modular design. However, high volumes of a sub-optimal design may serve better than low volumes of an optimized design, if the parts to build the optimized design in high volumes cannot be obtained in the required time.
Almost all external or chassis mount power supplies will have input and output connectors. Selecting industry standard connectors will help to enable locating sufficient inventory of suitable components from multiple manufacturers. Power supply and connector vendors can help to select appropriate industry standard connectors if the product design team is not familiar with them.
Specifying standard and common power supply voltages will help to increase the probability of available power supplies. Hopefully most engineers would recognize that 7.8 Vdc is neither a standard nor common supply voltage. However, it should also be recognized that although a section of the product load may require 9 Vdc and it may be a standard voltage that it is not as common as is 12 Vdc. Specifying a power supply module to provide 12 Vdc and then using either a modular linear or switching regulator to produce 9 Vdc for the section of the load requiring that voltage may make the components for the design easier to acquire.
Minimizing Future Supply Challenges
Although CUI, along with most other electric component vendors, would like to be able to deliver tens of thousands of any part requested by a customer within a few days or weeks, that is not a realistic expectation. The most efficient process to rapidly deliver large volumes of a product to the market is for the design teams to work with vendors early and through-out the design process to adopt achievable options.
You May Also Like
Have comments regarding this post or topics that you would like to see us cover in the future?
Send us an email at powerblog@cui.com




